Advanced oxidation, also known as deep oxidation, is based on the synergistic effects of light, electricity, catalysts, and oxidants to generate highly active free radicals in the reaction system. These free radicals can react with organic matter through addition, substitution, electron transfer, or by breaking chemical bonds, degrading refractory organic matter into low- or non-toxic small molecules. They can even directly degrade the matter into CO2 and H2O, achieving near-complete mineralization.
1. Introduction to Advanced Oxidation and Its Advantages and disadvantages
Common advanced oxidation methods include ozone oxidation, ozone catalytic oxidation, electrocatalytic oxidation, Fenton oxidation, and wet oxidation. Now we will introduce these four types of advanced oxidation methods.
1.1. Ozone oxidation
Ozone oxidation is achieved primarily through direct and indirect reactions. Direct reaction involves the direct reaction of ozone with organic matter, a highly selective method that typically targets organic matter with double bonds and is most effective against unsaturated aliphatic and aromatic hydrocarbons. Indirect reaction involves the decomposition of ozone to produce ·OH, which then oxidizes the organic matter through ·OH, a non-selective method.
Although ozone oxidation has a strong ability to decolorize and remove organic pollutants, the method has high operating costs, is selective in the oxidation of organic matter, cannot completely mineralize pollutants at low doses and in a short period of time, and the intermediate products generated by decomposition will hinder the ozone oxidation process.
Ozone has a high redox potential in water and is often used for disinfection, deodorization, deodorization, decolorization, etc. Ozone can oxidize many organic substances, such as proteins, amino acids, organic amines, chain unsaturated compounds, aromatics, lignin, and humus.
1.1.1. advantage
- Strong oxidizing ability and obvious effects on deodorization, decolorization, sterilization and removal of organic matter.
- The ozone in the treated wastewater is easily decomposed and does not cause secondary pollution.
- The air and electricity used to produce ozone do not need to be stored and transported, making operation and management more convenient.
- Generally no sludge is produced during the treatment process.
1.1.2. disadvantage
- Ozone is not easy to store and can only be used immediately after it is made.ozoneequipmentThe cost of ozone treatment is high, and the utilization rate is low, which makes the cost of ozone treatment high., the supporting factory buildings need to be explosion-proof, and the supporting pipes need to be treated with anti-corrosion.
- Ozone has a strong selectivity in its reaction with organic matter. It is impossible to completely decompose ozone at low doses and within a short period of time.mineThe decomposition of pollutants will inhibit the further oxidation of ozone.
- Cannot be effectively removedTotal nitrogen, has no oxidizing effect on organic chlorides in water.
- The higher the pollutant concentration, the greater the amount of ozone added.
1.2. Ozone catalytic oxidation
The efficiency of simple ozone oxidation is low. In order to improve the catalytic efficiency, catalysts are added to change ozone oxidation into ozone catalytic oxidation to improve the utilization efficiency of ozone.
Under the action of catalyst, after O3 dissolves in water, the following reaction occurs:
O3+H2O→O2+H2O2
H2O2+H2O→O2+2·OH
·OH can also induce a series of chain reactions, producing other ground-state substances and free radicals, which strengthens the oxidation effect. Depending on the form of the catalyst, it can be divided into homogeneous catalytic oxidation and heterogeneous catalytic oxidation.
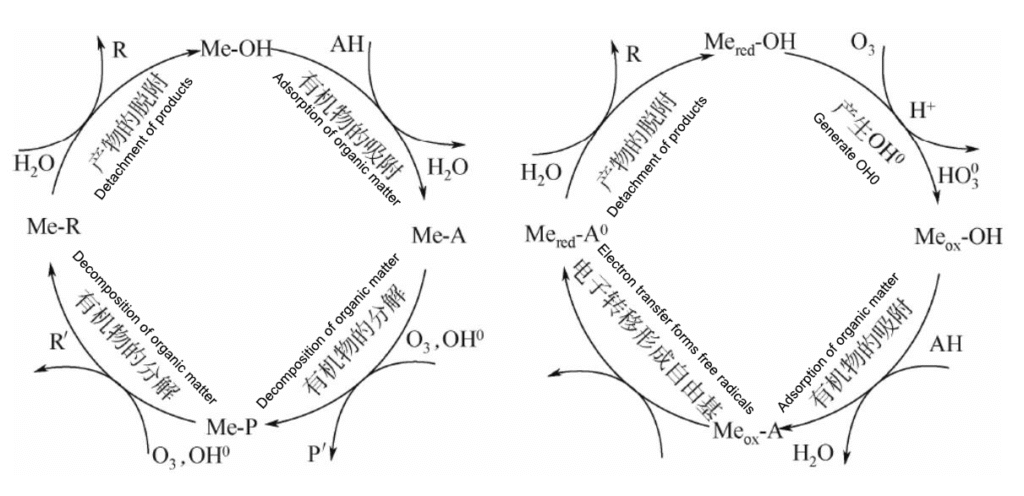
Schematic diagram of ozone catalytic oxidation principle
Using solid metals, metal oxides or metals or metal oxides supported on a carrier to carry out catalytic reactions;
In heterogeneous catalytic ozonation systems, there are generally three possible reaction mechanisms:
1) Ozone chemical adsorption on the catalyst surface leads to the formation of active substances, which react with non-chemically adsorbed organic molecules;
2) Chemical adsorption of organic matter on the catalyst surface and its further reaction with gaseous or liquid ozone;
3) Both organic matter and ozone are adsorbed on the catalyst surface and then react with each other at the chemical adsorption sites.
1.2.1. advantage
- Highly efficient degradation of pollutants: by catalyzing the production of hydroxyl radicals (·OH), the oxidation rate is 2-5 orders of magnitude higher than that of ozone alone, and it can degrade difficult-to-biodegrade organic matter (such as halogenated hydrocarbons and polycyclic aromatic hydrocarbons), some of which can be completely mineralized.
- No secondary pollution: The reaction products are CO₂ and H₂O, and no additional pollutants are introduced.
- Wide range of applications: It is suitable for the pretreatment and deep treatment of high-concentration, high-toxic, and difficult-to-degrade industrial wastewater (such as chemical, pharmaceutical, printing and dyeing, etc.). It can also be used for reverse osmosis concentrated water and membrane pollution control.
- Improve ozone utilization efficiency:Reduce ozone dosage and operating costs. Some processes can be combined with biological treatment to further reduce costs.
1.2.2. disadvantage
- Catalysts need to be added, but they are expensive and have the risk of loss and passivation, so they need to be replaced regularly.Catalyst (metal ion) recovery is difficult and may cause secondary pollution.
- The reaction temperature and pH value need to be controlled (some processes require an acidic environment), and high requirements are placed on the equipment material (ozone corrosion resistance)..
- Ozone needs to be produced and used immediately, which requires high equipment investment and energy consumption., the supporting factory buildings need to be explosion-proof, and the supporting pipes need to be treated with anti-corrosion.
- Treatment efficiency limitations: The treatment cycle for high-concentration COD wastewater is long, and it is difficult to quickly reach the standard when used alone., and reduce the service life of the catalyst.
1.3. electrocatalytic oxidation
Electrocatalytic oxidation technology is driven by an external electric field, inducing electron transfer reactions on the electrode surface, thereby generating highly oxidizing active substances such as hydroxyl radicals (·OH) and reactive oxygen species (ROS). These active substances can effectively oxidize and decompose organic matter into carbon dioxide (CO₂), water (H₂O), or other small molecules.
Specifically, the electrocatalytic oxidation process includes two main oxidation mechanisms:
Direct oxidation: Organic compounds lose electrons directly at the anode surface and are oxidized into intermediate or final products. This oxidation method depends on the properties of the anode material, such as titanium-based coated electrodes.
Indirect oxidation: Various oxidants are generated through electrolysis, such as chlorine (Cl₂), ozone (O₃), hydrogen peroxide (H₂O₂), etc., or reactive oxygen species (such as ·OH) adsorbed on the electrode surface are used to oxidize organic matter. Indirect oxidation not only improves oxidation efficiency but also expands the treatment range of organic matter. Electrochemical oxidation technology mainly focuses on the treatment of biotoxic and difficult-to-degrade aromatic compounds.
1.3.1. advantage
- High efficiency and pollution-free: No need to add chemical agents to avoid secondary pollution; it has strong oxidation ability and can degrade difficult-to-biodegrade organic matter.
- Mild operating conditions: Operate at normal temperature and pressureflexible.
- Multifunctional: It can simultaneously remove COD, ammonia nitrogen, and color, and also has a bactericidal effect.
- Small footprint:The equipment has high integration and is suitable for pretreatment or deep treatment of high-concentration wastewater.
1.3.2. disadvantage
- High energy consumption: The power consumption is significant when treating high-concentration wastewater, and the operating cost is high, lower when treating saline wastewater.
- Electrode loss:Long-term operation may cause electrode corrosion or catalyst deactivation, and regular maintenance is required.
- Cost constraints:Core catalytic electrodeThe high cost of precious metal catalysts (such as Pt and Ir) limits their large-scale application.
1.4. Fenton oxidation
The Fenton oxidation process uses Fe2+ to catalyze the decomposition of H2O2 under acidic conditions, producing ˙OH to degrade pollutants. The resulting Fe3+ then undergoes coagulation and precipitation to remove organic matter. Therefore, the Fenton reagent has both oxidation and coagulation effects in water treatment. The oxidation of organic matter occurs through the reaction of Fe2+ with H2O2, generating the highly oxidizing hydroxyl radical ˙OH. Furthermore, the resulting Fe(OH)3 colloid has flocculation and adsorption properties, potentially removing some organic matter from water.
Fenton’s reagent is a wastewater treatment method that uses ferrous ions (Fe2+) as a catalyst and hydrogen peroxide (H2O2) for chemical oxidation. It can generate highly oxidizing hydroxyl radicals, which react with refractory organic matter in aqueous solution to generate organic free radicals, destroying their structure and ultimately oxidizing and decomposing them to achieve the purpose of reducing the COD of the wastewater.
The Fenton oxidation reaction equation is roughly as follows:
Fe2++H2O2=Fe3++OH–+HO·
Fe3++H2O2+OH–=Fe2++H2O+HO·
Fe3++H2O2=Fe2++H++HO·
Refractory organic matter in wastewater undergoes coupling or oxidation to form smaller molecular weight intermediates, thereby altering their biodegradability, coagulation and sedimentation properties, and solubility. These intermediates can then be removed through subsequent coagulation and sedimentation or biochemical methods to achieve purification. This is the most commonly used advanced oxidation process, with a wide range of applications.
1.4.1. advantage
- Strong oxidizing ability: The hydroxyl radical (·OH) is generated by the reaction of Fe²⁺ with H₂O₂, which can efficiently degrade difficult-to-biodegrade organic matter (such as polycyclic aromatic hydrocarbons and halogenated hydrocarbons) and partially achieve complete mineralization.
- Mild operating conditions: Simple operating conditions,Pharmacy is common and easy to buy.Easy to operate, wide range of adaptability.
- Low investment: The process flow is simple, requiring only a mixing reaction tank and a sedimentation tank, and requiring little infrastructure investment.
- Widely used: It can be used alone as a pretreatment or deep treatment process, or in combination with other technologies.
1.4.2. disadvantage
- High consumption of reagent:A large amount of Fe²⁺ and H₂O₂ is required, which results in high operating costs, and excessive Fe²⁺ will lead to an increase in sludge volume., and introduced inorganic salts to increase TDS.
- Produce secondary pollution: Residual Fe²⁺/Fe³⁺ may form iron sludge, which requires additional treatment; residual H₂O₂ may affect the effluent quality.
- Reaction efficiency limitations: When used alone, the removal rate of high-concentration COD is limited, and the reaction time needs to be extended or combined with other processes., and the precipitation effect may be poor.
- pH Limitation: The optimal reaction pH is 2-4, and additional wastewater pH adjustment and additional treatment steps are required.
1.5. Wet oxidation
Wet oxidation technology refers to a chemical process in which organic pollutants are oxidized into inorganic substances such as carbon dioxide and water or small molecular organic substances in the liquid phase using oxygen in the air as an oxidant under high temperature and high pressure, including homogeneous wet catalytic oxygen and heterogeneous wet catalytic oxygen.
Wet oxidation technology can almost non-selectively oxidize various high-concentration organic wastewaters, especially those that are highly toxic and difficult to degrade using conventional methods. However, it is expensive and has demanding operating conditions. Currently, its main applications include industrial wastewaters such as papermaking wastewater, cyanide wastewater, and pesticides.
1.5.1. advantage
Compared with conventional methods, it has a wide range of applications, high treatment efficiency, very little secondary pollution, fast oxidation rate, and can recover energy and useful substances.
1.5.2. disadvantage
- Wet oxidation is generally required to be carried out under high temperature and high pressure conditions. The intermediate products are often organic acids, so the requirements for equipment materials are high. They need to be resistant to high temperature, high pressure, and corrosion. Therefore, the equipment cost is high and the one-time investment of the system is high.
- Since wet oxidation reactions need to be carried out under high temperature and high pressure conditions, they are only suitable for treating wastewater with small flow rates and high concentrations, and are not economical for treating wastewater with low concentrations and large volumes.
- Even at very high temperatures, the removal effect of certain organic substances such as polychlorinated biphenyls and small molecule carboxylic acids is not ideal, and it is difficult to achieve complete oxidation.
- During wet oxidation, highly toxic intermediates may be produced.





